Electro Chemistry Formulas
Electro Chemistry is one more important concept in Chemistry subject, it helps you understand the connection between electrodes and chemical reactions. You can get familiar with the concept by referring to the Electro Chemistry formulas. This Electro Chemistry formulae list is designed by subject experts by focusing on all important topics in the Electrochemistry chapter. In this formula sheet of Electro Chemistry, you can revise all crucial formulas like Faradays’s first law of electrolysis & second law, Kohlrausch’s law. So, make the most out of this Electro Chemistry Formula Tables, Sheets.
Solve your chemistry problems fastly and efficiently taking the help of Chemistry Formulas and learn about the Concepts without much effort.
Formula Sheet for Electro Chemistry
Make use of the Electro Chemistry Formulas and revise all the concepts thoroughly in a quick way. To memorize these Electro Chemistry Formulas use the cheat sheet and table prevailing here & get more support during your exam preparation. So, avail it from here and just master in the Electro Chemistry concept.
1. Table:
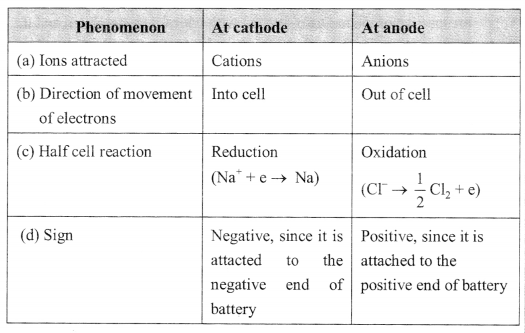
2. (i) Faradays’s first law of electrolysis:
“For the same electrolyle, the mass of a substance produced or
consumed at an electrode is directly proportional to the quantity of
charge (electricity) passed through the electrolytic cell”.
![]()
or m = Z.I.t & Z = \(\frac{E}{F}=\frac{E}{96500}\)
(ii) Faraday’s second law of electolysis:
“When same quantity of electricity is passed through different
electrolytes, the amount of products obtained are proportional to
their chemical equivalents or equivalent weights.”
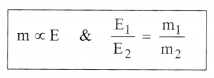
3. R = ρ\(\frac{\ell}{a}\) & G = \(\frac{1}{\mathrm{R}}\) & x = RK = \(\frac{\ell}{a}\)
4. \(\bar{\lambda}_{\mathrm{eq} .}=\frac{1}{\rho} \times
\mathrm{V}\)
& λmolar = \(\frac{1}{\rho}\) × Vm
& λm° – λmolar = b√C
5. Kohlrausch’s law:
“At infinite dilution, when dissociation is complete, each ion
makes a definite contribution towards equivalent conductance of the
electrolyte irrespective of the nature of the ion with which it is
associated and the value of equivalent conductance at infinite
dilution for any electrolyte is the sum of contributions of its
constituent ions.”
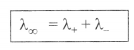
☞ Degree of dissociation:
α =
\(\frac{\lambda_{\mathrm{eq}}}{\lambda_{\mathrm{eq}}}=\frac{\text {
Equivalent conductance at a given concentration }}{\text {
equivalent conductance at infinite dilution }}\)
6. Nernst Equation:
E = E° – \(\frac{0.0591}{n} \log _{10} \frac{[\text { Pr
oducts }]}{[\text { Reactants }]}\)
& E°cell = E°right + E°left
& Keq =
antilog\(\left[\frac{\mathrm{nE}^{\circ}}{0.0591}\right]\)
ΔG = – nFEcell & ΔG° = – nFE° cell
& Wmax = + nFE° & ΔG = ΔH +
T\(\left(\frac{\partial \Delta \mathrm{G}}{\partial
\mathrm{T}}\right)_{\mathrm{P}}\)
Visit chemistrycalc.com a one-stop destination for all Chemistry concepts formulas along with Electro Chemistry Formula Sheets and Tables.