Iron III Oxide Formula
Iron III Oxide
Iron III Oxide Formula- It is one of the main oxides of iron and is an inorganic compound. Besides, the other two compounds are iron ii oxide (FeO) which is rare, and the other one is iron (II, III) oxide (
Iron III Oxide Formula and Structure
Its chemical formula is
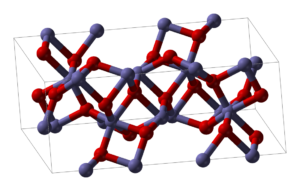
Alpha Phase
The alpha-iron iii oxide or ferric oxide has the rhombohedral, corundum (
We can prepare it using precipitation and thermal decomposition in the liquid phase. However, its magnetic properties depend upon particle size, pressure, and magnetic field intensity.
Gamma Phase
The gamma-ferric oxide has a cubic structure. In addition, it is metastable and converted from the alpha phase at high temperatures. Naturally, it occurs as the minerals maghemite. This phase is ferromagnetic and we use it in recording tapes, while ultrafine particles smaller than 10 nanometers are superparamagnetic.
Occurrence of Iron III Oxide
Naturally, it occurs in many forms such as hematite, and maghemite. Furthermore, we can synthetically produce it by igniting virtually any ferrous compounds in the air. It also forms a number of hydrates with adaptable compositions and structures.
Preparation of Iron III Oxide
The ferric oxide or iron iii oxide is a product of the oxidation of iron. In laboratories they prepare it by electrolyzing a solution of sodium bicarbonate in the inert electrolyte with an iron anode:
This subsequent hydrated iron iii oxide written here as FeO(OH), dehydrates around
Properties of Iron III Oxide
It is not conducive to electricity but some perovskite is electrically conductive. They are soluble in acids but are not soluble in water or all convention organic solvents. Its density is
Uses of Iron III Oxide
Majorly, industries use it as the feedstock of the steel and iron industries such as for the production of steel, iron and many alloys. Furthermore, jewelers use its fine powder that we call jewelers’ rouge, or red rouge as a final polish on metallic jewelry and lenses and traditionally as cosmetics. In addition, it cuts more slowly than some modern polishes such as cerium (IV) oxide.
Industries also use it as a pigment under names ‘pigment brown 6’ ‘pigment brown 7’ and ‘pigment red 101’. Moreover, the dentist uses them in dental composites together with titanium oxides. It is the most common magnetic particle that they use in all types of magnetic storage and recording media that include magnetic disks and magnetic tapes.
Solved Example for You
Question: Mention a reaction of iron iii oxide?
Solution: The reaction is:
This is one of the most important reactions in carbothermal reduction that gives iron that we use in the making of steel and it releases carbon dioxide.