Lithium Oxide Formula
Lithium Oxide
Lithium Oxide Formula, we also know it by the name Kickerite or lithia. It is a highly insoluble thermally stable source of lithium suitable for making glass, ceramics, and optics. It forms when lithium metal burns in the presence of oxygen. Furthermore, these compounds (oxides compounds) are not conductive to electricity. But some perovskite-structured oxides are electrically conductive and finding application in the cathode of solid oxides fuel cells and oxygen generation systems. Moreover, it is an inorganic compound.
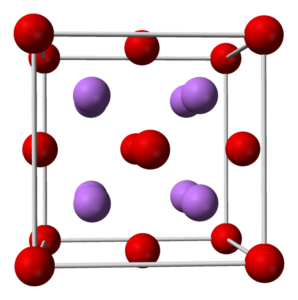
Lithium Oxide Formula and Structure
Its chemical formula is
In addition, the gas phase
Preparation of Lithium Oxide
We can prepare lithium oxide by the thermal decomposition of lithium peroxide at
In addition, we can produce pure
Properties of Lithium Oxide
It is a white crystalline solid and its density is
Also, it is soluble in water and when we dissolve it in water it forms lithium hydroxide. Its refractive index is 1.644 and the crystals structure is cubic anti-fluorite, cF12. In addition, its heat capacity is 1.8105 J/g K or 54.1 J/mol K.
Uses of Lithium Oxide
We use lithium oxide in ceramic glazes as a flux, and for creating blues with copper and pinks with cobalt. Furthermore, it reacts with steam or water, forming lithium hydroxide and should be isolated from them.
Inside the thermal barrier coating, its usage is also being investigated for non-destructive emission spectroscopy evaluation and degradation monitoring.
Moreover, we can add it as a co-dopant with yttria (Yttrium oxide which is a white solid and air-stable substance) in the zirconia ceramic topcoat, exclusive of a large decline in the predictable service life of the coating.
In addition, at high temperature or heat, lithium oxide emits a very detectable spectral pattern that expands in strength together with degradation of the coating.
Most noteworthy, its implementation would allow in situ monitoring of such systems, enabling an efficient means to protect lifetime until failure or necessary maintenance. However, we can obtain lithium metal from lithium oxide by the electrolysis that releases oxygen as a by-product.
Safety and Health Hazards of Lithium Oxide
It is very corrosive and reacts violently with water to form lithium hydroxide. Buring it produces toxic fumes that if inhaled can cause irritation in the respiratory tract, and if it comes in contact with the skin then it causes irritation in the eyes, nose, and skin.
Besides, lithium oxide is toxic because of its strong alkalinity. Besides, long exposure to skin and eyes can cause severe burns and blindness. Also, it can damage the central nervous system. The initial effect of exposure include shaking of hands, micturition, slurred speech, nausea, sluggishness, sleepiness, vertigo, thirst and increased volume of urine.
Solved Example for You
Question: Mention the simplest method to prepare lithium oxide?
Solution: The simplest method to prepare lithium oxide is as follows: