Oxalate Formula
Incomplete oxidation of carbohydrates in the plants leads to the synthesis of oxalate compounds. Many metal ions, while reaction with the oxalates are forming insoluble precipitates. Oxalates are also capable of forming the coordination compounds. Calcium oxalate is a much common salt of this kind. The primary constituent of the most common kind of oxalate may be in the human body in the form of kidney stones. This article will discuss the Oxalate formula with the examples. Let us learn it!
Source: en.wikipedia.org
Oxalate Formula
What is Oxalate?
Oxalates are the salts of oxalic acid with certain metal ions. These are under natural conditions and are not toxic. But becomes toxic because of the oxalate ion. Oxalates are the dicarboxylic acid di-anion. Its name is often used for derivatives like salts of oxalic acid. For example sodium oxalate or dimethyl oxalate.
The formula for Oxalates
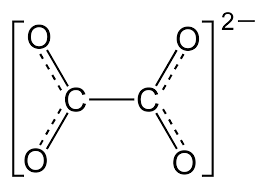
The formula for the oxalate ion is as follows
Or
It is also called Ethanedioate or Oxalate Ion. One can obtain it by deprotonation of both the carboxyl groups of
Properties of Oxalate Ion
Oxalate ions are planar in some crystalline oxalates. Ethanedioate is easily accessible in many plants. Its formation is possible due to the incomplete oxidation of the carbohydrates.
The roots and leaves of buckwheat and rhubarb are some popular and common examples of oxalate-rich plants. It plays the role of being a plant metabolite or a human metabolite. It has 4 hydrogen bond acceptors.
With the help of X-ray crystallography, we can see its simple structure which appears as a planar conformation with symmetry.
Uses of Oxalate Ion
- Some Oxalate like calcium oxalate is useful in the manufacturing of ceramic glazes.
- Escitalopram Oxalate is useful in the treatment of anxiety and depression.
- In the powder form, it is useful as a pesticide in beekeeping.
- It acts as an excellent ligand for metal ions.
Health hazards of Oxalates
Oxalic acid in the human body combines with the divalent metallic cations like iron (II) and calcium to produce crystals of the corresponding oxalates. Then we can excrete these crystals from urine as minute crystals.
Oxalates may vary in the form of larger kidney stones which can obstruct the kidney tubules. Approximately 80 % of the kidney stones are only due to calcium oxalate salt.
Solved Examples
Q.1: Is oxalate a solid?
Solution: These crystals may stick together and form a solid mass (a kidney stone). Oxalate is one type of compound that may form the crystals in the urine.
Q.2: Is sodium oxalate an acid or base?
Solution: Sodium oxalate is common and basic salt. According to the Bronsted and Lowry theory of acids and bases, this oxalate ion is a conjugate base. We may derive it from the oxalic acid. As it is the conjugate base of hydrogen oxalate
Q.3: Determine the molar mass of Oxalate ion.
Solution: Formula for Oxalate ion is:
Atomic mass of C = 12.0107
Atomic mass of O = 15.999
Its molar mass will be,
=
= 88.02 gram per mole.
Therefore the molar mass of oxalate ion will be 88.02 gram per mole.