Charle's Law Calculator
Take the help of free Charle's Law Calculator to determine ideal gas parameters in an isobaric process. You just have to enter the known volume, temperature details in the input fields of the calculator and click on the calculate button to avail the result.
Charle's Law Definition
Charle's law states that the volume of gas for a fixed mass is directly proportional to the temperature. Sometimes, it is also called the law of volumes. t defines the relationship between volume and temperature of a gas at constant pressure.
Charles law is defined as the ratio of volume and temperature of a gas in a system that remains constant when the pressure is constant. The law indicates the behaviour of an ideal gas in an isobaric process.
Charle's Law Formula & Derivation
According to the Charles law, the equation is V ∝ T.
V₁/T₁ = V₂/T₂
Where,
- V₁ is the initial volume
- T₁ is the initial temperature
- V₂ is the final volume
- T₂ is the final temperature
The formula to compute the final volume is V₂ = V₁ / T₁ * T₂
Final temperature is T₂ = T₁ / V₁ * V₂
Initial Volume V₁ = V₂ / T₂ * T₁
Initial Temperature T₁ = V₁ / V₂ * T₂
As per Charles law, the volume of gas is directly proportional to the temperature at constant pressure.
The statement is V∝T
So, V/T = constant = k
Let us take V₁ is the initial volume, T₁ is the initial temperature
The equation is V₁/T₁ = k --- (i)
If V₂ is the final volume, T₂ is the final temperature then equation is V₂/T₂ = k --- (ii)
By equating both equations
V₁/T₁ = V₂/T₂
Example:
Question: A gas occupies 220 cm3 at a temperature of 0 c and pressure of 760 mm Hg. What will be the volume at 110 C?
Answer:
Given that,
Initial volume V₁ = 220 cm3
Initial temperature T₁ = 0 c = 0 + 273 = 273 k
Final temperature T₂ = 110 C = 110 + 273 = 383 K
Charle's law formula is V₁/T₁ = V₂/T₂
Final volume V₂ = V₁/T₁ * T₂
= (220/273) * 383
= 308.64
Therefore, final volume is 308.64 cm3
Chemistrycalc.Com has got concepts like bio chemistry, physical chemistry, stoichiometry and solutions and many more calculators at one place.
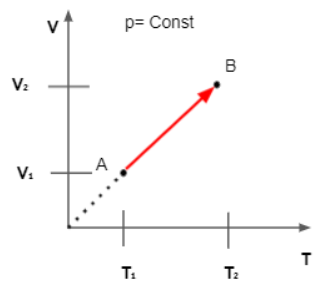
Charle's Law Graph
The following graph between the temperature and volume of a gas at constant pressure is called the Isobar or Isoplestics and it is always a straight line. The graph of volume versus temperature at constant pressure is shown below temperature 273.15°C and it is the lowest possible temperature.
.png)
Applications of Charle's Law
Some of the applications of Charle's law are provided here.
- Thermometer: It is a device that is based on the principle of Charle's Law. But, it has certain limitations like resistance to high temperatures and so on.
- Balloon Flight: Have you ever wondered by seeing the ballon flying in the sky? It is because of Charles law i.e whenever the air gets heated volume of the gas increases and density decreases. Bouyancy of Surrounding does the rest of the job for you and balloon starts floating.
- Liquid Nitrogen Experiments: If you place a balloon in a container that is filled with liquid nitrogen then it begins to shrink and freed no matter how it is in the initial phase.
Frequently Asked Question's on Charle's Law Calculator
1. What is Charle's Law Equation?
The Charle's Law formula is V₁ * T₂ = V₂ * T₁ at constant pressure. Here, V₁, T₁ are initial volume and temperature, V₂, T₂ are final volume and temperature.
2. What is Charle's Law?
Charles law states that the volume of gas for a fixed mass is directly proportional to the temperature. In other words, if the gas expands then the temperature is increased.
3. How to calculate Charles Law?
You have to enter the known gas parameters such as initial, final volume and temperature and press the calculate button to get the result easily.
4. What are the applications of Charle's Gas Law?
The daily life applications of Charles law are listed here.
- In the winter season, the capacity of the human lung decreases.
- In cold weather, the helium balloons shrink.