Cell EMF Calculator
Electromotive Force of a Cell Calculator is used to calculate the galvanic cell EMF by taking anode, cathode details. Just you need to enter the potential of the cathode, the potential of the anode in the specified input sections and press the calculate button to find the EMF of a cell effortlessly.
How to Calculate EMF of a Cell?
The simple steps to find the electromotive force of the cell are mentioned here. Follow these rules to get the output in a less amount of time.
- Get the potential of the cathode, potential of the anode of the cell.
- Subtract cathode from the anode to get the cell EMF.
Electromotive Force Sources
The some of the examples of EMF sources are listed-here:
- batteries
- alternators
- galvanic cells
- solar cells
- fuel cells
- thermocouples
- some living organisms (e.g., electric eel)
Each and every EMF source have an energy source that indues the flow of electric charges. All these sources may be the chemical processes, mechanical forces, temperature difference and electromagnetic radiation emitted by the sun.
Electromotive Force | EMF Equation
EMF or electromotive force is a force or an electric pressure which generates a current in a circuit. In simple words, EMF is the potential difference between the two electrodes of a cell. The unit of EMF is volts. The EMF of a galvanic cell specifies the ability of an electrochemical cell to cause the flow of electrons through the electric circuit.
The EMF equation is EMFcell = Ecathode - Eanode
Where,
EMF is the electromotive force
Ecathode is the potential of the cathode
Eanode is the potential of the anode
An electrochemical cell is a device that produces electrical energy from a chemical reaction. It is a type of energy source. It can be created by placing the metal electrodes in an electrolyte where chemical reaction wither uses or generates the current. It is also called the voltaic cells.
The different types of electrochemical cells are galvanic cells, Daniell cells. The Daniel cell is invented in 1836 by John Frederic Daniel. In this Daniel cell, copper is immersed in a solution of copper(II) sulfate and Zinc immersed in zinc sulfate solution.
Electropotential of Anode and Cathode
You can calculate the potential of a metal electrode in two methods. One is to check the galvanic series and find the particular metal in the below-given table. Another is using the Nernst equation also called the cell potential equation.
The Nerst equation is Ecell = Eocell - (RT)/(nF) logQ.
The electro potential of the metals table is as follows:
| Electrode | E⁰ [V] |
|---|---|
| Li / Li⁺ | -3.04 |
| Ca / Ca²⁺ | -2.86 |
| Mg / Mg²⁺ | -2.36 |
| Al / Al³⁺ | -1.69 |
| Mn / Mn²⁺ | -1.18 |
| Zn / Zn²⁺ | -0.76 |
| Cr / Cr³⁺ | -0.74 |
| Fe / Fe²⁺ | -0.44 |
| Cd / Cd²⁺ | -0.40 |
| Co / Co²⁺ | -0.28 |
| Ni / Ni²⁺ | -0.26 |
| Sn / Sn²⁺ | -0.14 |
| Pb / Pb²⁺ | -0.14 |
| Fe / Fe³⁺ | -0.04 |
| H₂ / 2H⁺ | 0.00 |
| Bi / Bi³⁺ | +0.32 |
| Cu / Cu²⁺ | +0.34 |
| Ag / Ag⁺ | +0.80 |
| Hg / Hg²⁺ | +0.85 |
| Au / Au³⁺ | +1.52 |
Electromotive Force Sources
The some of the examples of EMF sources are listed-here:
- batteries
- alternators
- galvanic cells
- solar cells
- fuel cells
- thermocouples
- some living organisms (e.g., electric eel)
Each and every EMF source have an energy source that indues the flow of electric charges. All these sources may be the chemical processes, mechanical forces, temperature difference and electromagnetic radiation emitted by the sun.
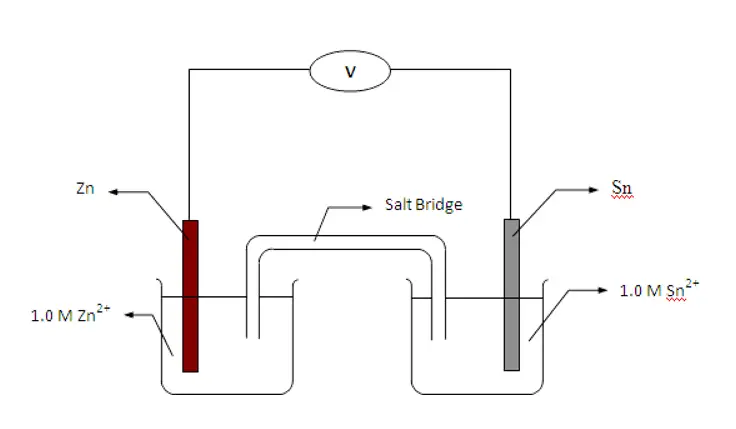
Galvanic Cell | Redox Reaction
The galvanic cell is invented by Luigi Galvani an Italian Scientist. It is an important electrochemical cell that forms the base of many cells. It has two metallic conductors i.e electrodes immersed in the ionic solutions. Each of these arrangements is a half cell. A salt bridge combines these two cells chemically. It gives electrons to the electron-deficient half cell and takes electrons from the electron-rich half cell. Here cathode is an electrode that has higher electric potential, the anode has the lower electric potential.
Example:
Question: Calculate the EMF of the redox reaction Mg(s) + 2H+(aq) → Mg2+(aq) + H2(g).
Answer:
Given redox reaction is g(s) + 2H+(aq) → Mg2+(aq) + H2(g)
Break the reaction into oxidation and reduction
2H+ + 2e- → H2
Mg → Mg2+ + 2e-
Find the standard reduction potential of the half-cell reaction
E0(reduction) = 0.0000 V
E0(oxidation) = -2.372 V
E0(oxidation) = -E0(reduction)
= 2.372
E0(cell) = E0(reduction) + E0(oxidation)
= 0.0000 + 2.372
= 2.372
Therefore, the EMF of the redox reaction is 2.372 V
If you would like to learn more about the other chemistry calculators that gives instant results, stay tuned to Chemistrycalc.Com
FAQ’s on Cell EMF Calculator
1. How to find the cell EMF without using a calculator?
The electromotive force of a galvanic cell can be calculated by subtracting the potential of the cathode from the potential of the anode.
2. What is the standard EMF of a cell?
The standard electromotive force of a cell is 1.100 v in an ideal solution.
3. What is an electrochemical cell and its types?
The electrochemical cell is a device that converts chemical energy into electrical energy. A chemical reaction that involves the exchange of electrons is needed for an electrochemical cell to operate. That reaction is called a redox reaction. The different types of electrochemical cells are galvanic cell, Daniell cell, electrode potential.
4. What is the Nernst equation for the electromotive force of a cell?
The Nernest equation is used for the determination of cell potential under non-standard conditions. The equation is Ecell = Eocell - (RT)/(nF) logQ
Ecell = Eocell - (0.0591)/ logQ at 25°C.