Michaelis-Menten Equation Calculator
Make use of the Michaelis-Menten Equation Calculator to discover the kinetics of a one-substrate reaction catalyzed with an enzyme. It is helpful to calculate the Michaelis-Menten constant, reaction rate, the concentration of the substrate, and the maximum rate of reaction. All you need to do is provide Michaelis constant, maximum reaction speed, substrate concentration details and click the calculate button to check the reaction rate as result.
How to Calculate Michaelis Constant Km?
Have a look at the simple steps to calculate the Michaelis Menten constant using the Menten equation Michaelis.
- Obtain the maximum reaction rate, substrate concentration, the reaction rate.
- Multiply the maximum reaction speed with the concentration of the substrate.
- Again multiply the reaction rate with the substrate concentration.
- Subtract the second product from the first product.
- Divide the result by the reaction rate to get the Michaelis Constant.
Michaelis-Menten Equation
The Michaelis-Menten Equation to find the reaction rate of an enzyme is given here:
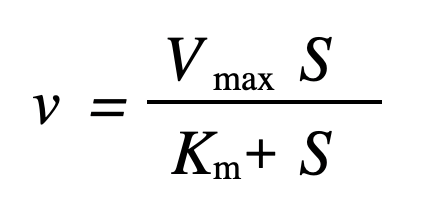
Where,
V is the rate of reaction
Vmax is the maximum reaction speed
[S] is the substrate concentration
Km is the Michaelis constant
The Michaelis-Menten is based on the kinetic concept, it can be expressed in the enzyme equation
E + S ⇄ ES → E + P
Here,
E is the Enzyme
S is the Substrate
P is the product
When an enzyme binds with the substrate, it results in a product. The enzyme can bind or unbind with various substrates back and forth, multiple times, that is why this reaction can read in two ways. Here, three enzyme kinetic equations in one
- E + S → ES, this is k1
- ES → E + S, this is k2
- ES → E + P, this is k3
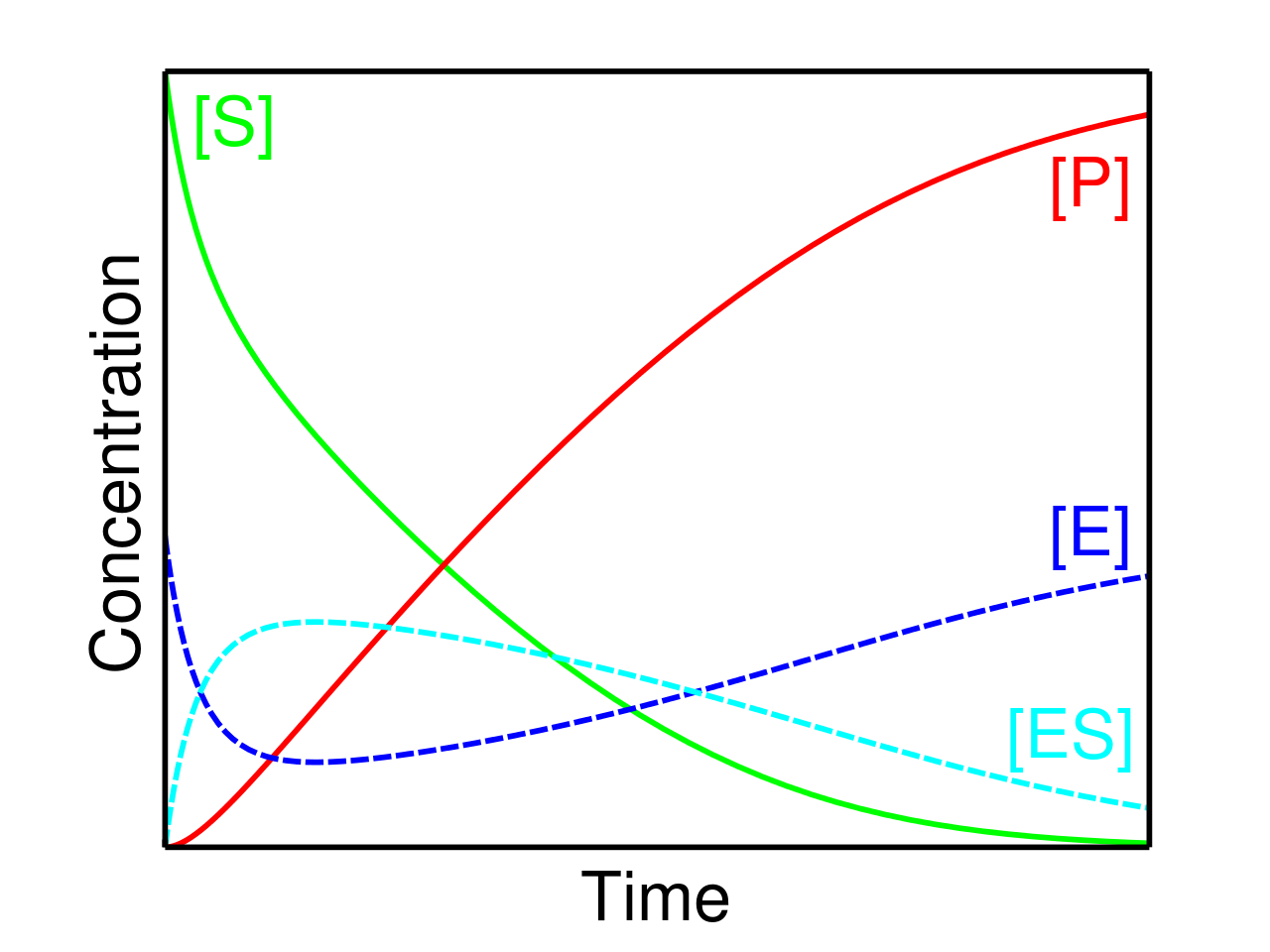
The amount of ES remains stable during the course of a reaction until the substrate is close to 0. The important observations of the Michaelis-Menten equation are as follows:
- If the substrate concentration is low, then the speed of the equation is directly proportional to the substrate.
- If the level of the substrate is high, then the speed is an independent parameter and it does not exceed the maximum rate.
Example:
Question: For a given enzyme catalyzed reaction, the Michaelis constant is 0.5 M, the concentration of substrate is 1.0 M, and the maximum reaction speed is 1.2 1/s. Find the reaction rate of the enzyme under these conditions.
Answer:
Given that,
Michaelis constant is 0.5 M
Substrate concentration is 1.0 M
Maximum reaction speed is 1.2 1/s
Michaelis-Menten Equation is V = (Vmax * [S]) / ([S] + Km)
V = (1.2 x 1.0)/(1.0 + 0.5)
= 1.2/1.5
= 0.8 1/s
Therefore, the reaction rate is 0.8 1/s.
At Chemistrycalc.Com, you will discover various concepts calculators like physical chemistry, stoichiometry, bio chemistry, and many more that assist you to make your calculations quickly and simply.
FAQ’s on Michaelis-Menten Equation Calculator
1. What are the limitations of the Michaelis-Menten Equation?
Michaelis-Menten Equation is the rate equation for one substrate for one enzyme-catalyzed reaction. This related the initial reaction rate, maximum reaction rate, initial substrate concentration through Michaelis constant. We have to make various assumptions to derive the Michaelis-Menten equation. The catalytic step is slower, so the rate is limiting.
2. What is the unit of Km and Vmax?
The unit of Michaelis-Menten constant Km is molar and the maximum reaction speed Vamx is 1/s.
3. What is the Michaelis Menten equation?
The Michaelis-Menten equation is V = (Vmax x [S])/([S] + Km). Here, V is the reaction rate, Vmax is the maximum reaction rate, [S] is the substrate concentration, Km is the Michaelis constant.
4. What factors can influence the Michaelis-Menten constant?
Michaelis constant Km is the amount of substrate present when the reaction rate is at half of the maximum. The factors that affect the Michaelis-Menten constant Km are pH, temperature, ionic strengths, and the nature of the substrate.

