Electron Dot Formula
Electron dot formula shows the number of valence electrons for that element with the help of dots. The valence electrons are those electrons that occupy the highest energy level. We can obtain it by using the periodic table. For example, the elements in group IA of the chemical periodic table have 1 valence electron. In chemistry electron dot formula has its own importance. This article will explain the electron dot formula with examples. Let us learn it!
Electron Dot Formula
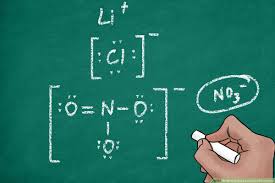
Concept of Electron Dot Formula
We also know the Electron dot structures as Lewis dot formula. We can draw it if we know the molecular formula of the compound. It is also useful to represent the atomic state of elements. We represent an electron dot structure by using the element symbol. Start from the top of the element symbol, then add dots in a clockwise manner to complete the number of valence electrons. The maximum number of electrons can be eight.
It defines the nature of the bond and position of atoms of the molecule which are connected in the molecule. The representation of molecules in Lewis electron dot structure is in the honor of the American chemist G N Lewis. Electron dot structures are the diagrams to describe the chemical bonding between atoms in a molecule. They also display the total number of lone pairs available in each of the atoms of the molecule.
How to Draw Electron Dot Formula?
Electron Dot Formula comprises of one dot for every valence electron with the element’s symbol. Note down that a skeletal structure displaying a realistic bonding pattern by means of only the element symbols is very important.
The steps needed to draw them are:
- First, calculate the total number of valence electrons present in the molecule by adding the individual valences of each atom.
- If the molecule is an anion, then we need to add extra electrons to the dot structure.
- If the molecule is a cation, then we need to subtract electrons from the total count.
- The least electronegative atom is at the central atom of the molecule or ion.
- The atoms are now connected with the single bonds.
- Now, the lone pairs of electrons are assigned to each atom in the molecule.
- After this, if every atom does not have an octet configuration, then a double or triple bond should be used to satisfy the octet valency of each atom.
- At last, if required, we have to convert a lone pair into a bond pair in order to satisfy the octet rule for two atoms
Solved Examples on Electron Dot Formula
Q.1: Explain the Electron Dot Formula of
Solution:
- The central atom of this molecule is the carbon atom.
- Oxygen contains 6 valence electrons with 2 lone pairs. Then, it is bonded to only one carbon atom with a double bond.
- Carbon contains four valence electrons, therefore giving zero lone pairs. So, it is doubly bonded to each oxygen atom.
Q.2: Give the electron dot formula for Aluminum atom.
Solution:
Aluminum is in group IIIA of the chemical periodic table. So, it has three valence electrons. So, its symbol Al will be surrounded by three dots.
Q.3: How do electron dot formulas represent shared electrons?
Solution: In the Electron dot formula, we denote the electrons by dots around the symbol of elements. Electrons sharing for covalent bonding is the glue for holding the atoms together.