Nitrogen Dioxide Formula
This article will explain Nitrogen Dioxide and its formula. This gas is an extremely poisonous chemical. It can be useful as an inhibitor. It is a pollutant that absorbs UV light and hence does not allow it to come to the surface of the earth. This chemical compound is paramagnetic in nature and it contains two oxygen atoms connected to one nitrogen atom. Students will learn the Nitrogen Dioxide formula with examples. Let us begin it!
Source: en.wikipedia.org
Nitrogen Dioxide Formula
What is Nitrogen Dioxide Gas?
It is also termed as Nitrogen oxide or Deutoxide of nitrogen. It is one of the major atmospheric pollutants which helps to absorb UV lights. This oxide is a yellowish-brown liquid in its compressed form or reddish-brown gas form. Its vapors are much heavier in comparison to air.
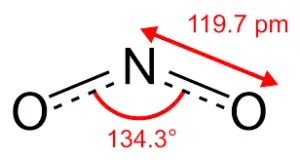
This chemical has the bond angle
The formula for Nitrogen Dioxide
Its chemical molecular formula is :
It has one N atom in bonding with 2 O atoms. Its density is 1.880 gram per cubic dm, Its boiling point is
Some common uses of NO2
- Nitrogen dioxide is useful as an intermediate in the production of nitric acid.
- It is useful in the manufacturing of oxidized cellulose compounds.
- It is useful as a catalyst and as an intermediate in the production of sulfuric acid.
- Also, it is useful as an oxidizer for rocket fuels as well as a nitrating agent.
- It is useful as an oxidizing agent.
- Nitrogen dioxide is also useful in the making of explosives.
Some Chemical Properties of Nitrogen dioxide
- It exists in an equilibrium state with dinitrogen tetroxide gas. Its equation is:
- Due to the weakness of the N – O bond, this chemical is a strong oxidizer.
- Hydrolysis reaction produces nitrous acid with nitric acid. Its equation is:
- It is a negligibly slow reaction at low concentrations of nitrogen dioxide.
- Corresponding nitrites are formed by alkyl and metal iodides.
Solved Examples
Q.1: Why Nitrogen Dioxide is useful. Find out its molar mass.
Solution: Nitrogen dioxide is the chemical compound and it is is one of several oxides of nitrogen. It is an intermediate in the industrial synthesis of nitric acid, millions of tons of which are produced each year. It is primarily useful in the production of fertilizers.
Its Chemical formula is:
So, its molar mass will be:
The atomic mass of
=
=46.0055 gram per mole
Thus the molar mass of it is 46.0055 gram per mole.
Q.2: What are the health hazards issues of Nitrogen Dioxide?
Solution: Severe exposures of Deutoxide of nitrogen is very much fatal. When in contact it may cause a burning sensation to eyes and skin. When it is in liquid form then it causes frostbite. It is reported to react with the blood to form chemical methemoglobin. When it was heated then it decomposes and hence releases toxic fumes of nitrogen oxides.